By Maurizio Macaluso, MD, DrPH, FACE, co-principal investigator with the Rare Diseases Clinical Research Network’s Data Management and Coordinating Center; professor and director of the Division of Biostatistics and Epidemiology at Cincinnati Children’s Hospital Medical Center; and professor in the Division of Pediatrics at the University of Cincinnati College of Medicine.
A preliminary analysis of more than 3,400 responses to a recent survey of people living with rare diseases and their caregivers is shedding new light on their experiences during the novel coronavirus (COVID-19) pandemic.
While only 71 responses indicated a positive COVID-19 diagnosis, a significant number of respondents were uncertain of their COVID-19 infection status due to difficulties accessing testing and medical advice. Most who reported COVID-19 infection experienced a mild infection, with serious interventions (e.g., mechanical ventilation) reported only by a few. Many reported some interaction between the infection and their rare disease, with one increasing the severity of the symptoms of the other.
The survey responses also indicated that the pandemic negatively affected rare disease patients in terms of access to regular health care, treatment for the rare disease, special diet, and special treatment and hospitalization, even among those who reported acquiring the infection.
An Update: RDCRN Survey on Impacts of COVID-19 on Rare Disease Patients and Families from Rare Diseases Network (RDCRN) on Vimeo.
The survey was launched in May 2020 by the Rare Diseases Clinical Research Network (RDCRN) with support from the National Institutes of Health’s National Center for Advancing Translational Sciences (NCATS) and the RDCRN’s 20 disease-specific consortia or research groups. The goal was to help the rare disease research community shed light on the needs of people with rare diseases and their caregivers during the COVID-19 pandemic and other potential health crises, in addition to informing future research efforts.
Preliminary data analyses
Results presented here reflect the 3,413 survey responses submitted through the closing date of December 15, 2020.
Individuals from the entire country participated in the survey, with 26 states contributing 50 or more surveys. Participants were predominantly women (64%) and white (89%). Minorities were under-represented in all subgroups of rare diseases. About 80% of the participants were adults.
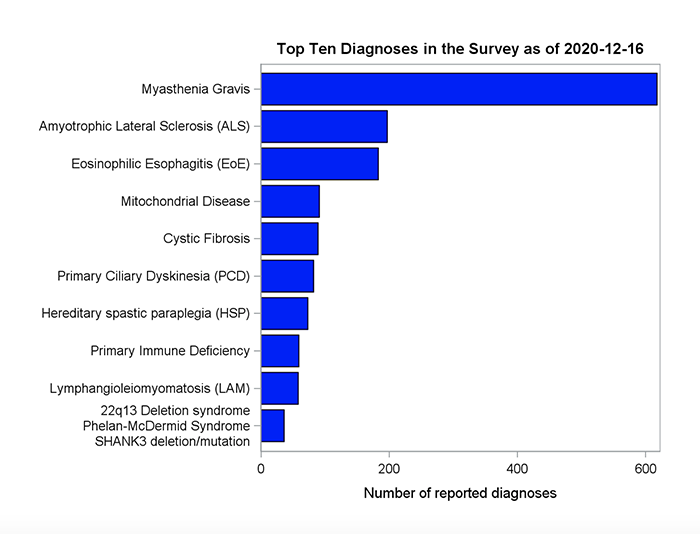
More than 130 distinct rare disease diagnoses were reported by respondents. The five most common diagnoses were myasthenia gravis, amyotrophic lateral sclerosis, eosinophilic esophagitis, mitochondrial disease and primary ciliary dyskinesia.
While 71 respondents reported acquiring a confirmed COVID-19 infection, 566 respondents (17% of the total) were not sure if they acquired the infection.
Characteristics of the 71 rare disease patients who reported a diagnosis of COVID-19 were:
- Mostly female (75%), white non-Hispanic (83%), adult (86%)
- Seen at emergency dept. or urgent care center: 28 (39%)
- Hospitalized: 7 (10%)
- Required oxygen: 12 (17%)
- Required mechanical ventilation: 4 (6%)
- Received investigational drugs: 9 (13%)
- Mean duration of symptoms: 27 days (range: 1-180)
- Symptoms not yet resolved at the time of survey: 24 (37%)
- Rare disease complicated COVID illness: 30 (44%)
- Rare disease symptoms worsened by COVID illness: 39 (55%)
The experience of those who reported COVID-19 infection was mostly that of a mild infection, with serious interventions (e.g., mechanical ventilation) reported only by a few.
Many reported some interaction between the infection and their rare disease, with one increasing the severity of the symptoms of the other.
The following chart shows reported COVID-19 infection status by RDCRN consortium. Those who acquired COVID-19 are shown in blue; those who did not acquire COVID-19 are shown in orange; those unsure of a diagnosis are shown in gray.
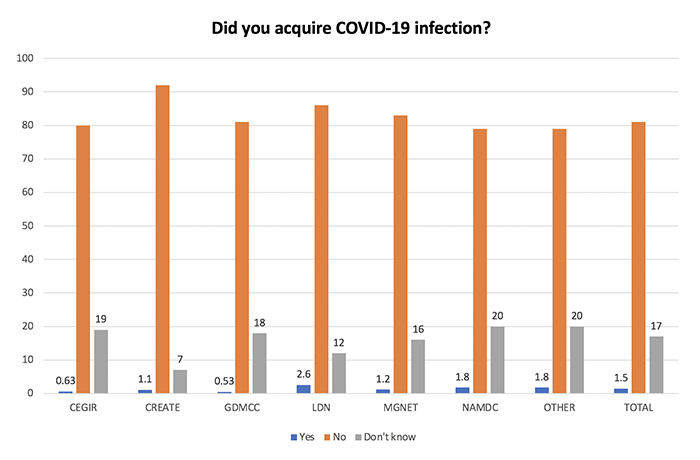
Obtaining access to testing/advice on COVID-19 was more difficult among those who did not know if they acquired COVID. This finding raises concerns that the infection may have been underdiagnosed in the RD community.
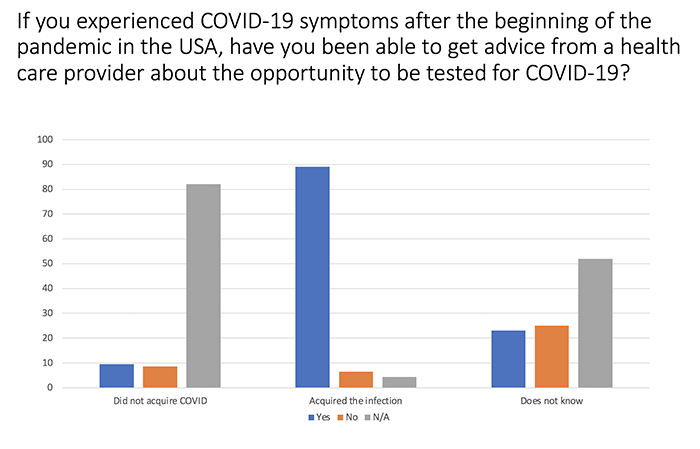
We found that survey participants who reported that they had acquired Covid-19 had increased cough, fever greater than 100.5, new or increased shortness of breath, headache, loss of taste and confusion much more often than participants who reported that they had not acquired Covid-19.
Those with unsure infection status often reported symptoms consistent with COVID-19 infection, reinforcing suspicion that COVID-19 infection was underdiagnosed.
Impact of the Pandemic
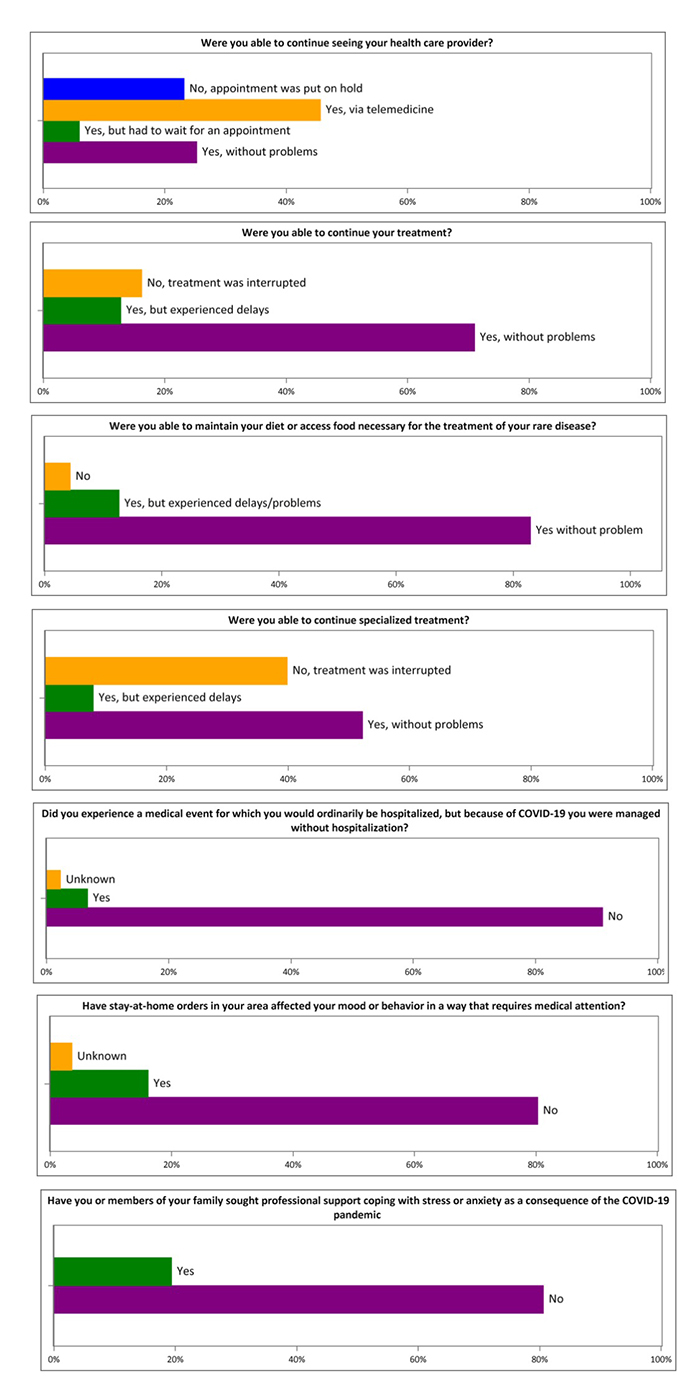
The survey responses indicated that the pandemic negatively affected rare disease patients and their caregivers in terms of access to regular health care, treatment for the rare disease, special diet, and special treatment and hospitalization, even among those who reported acquiring the infection. Some respondents had difficulty receiving treatments, especially those requiring special diets, occupational therapies, and physical therapies. The pandemic also caused mood changes, anxiety and stress in both the patients and their family members to an extent that required medical attention.
Next steps
This survey is the first of many initiatives being planned by the RDCRN and its partners to respond to the COVID-19 pandemic and its impacts on the rare disease community. As next steps:
- An RDCRN workgroup is planning additional analyses. The team will also develop a publication plan and evaluate and coordinate requests for access to data in a shared analysis environment in the NCATS Cloud.
- RDCRN is collaborating with the National Institute of Allergy and Infectious Diseases and other institutes at NIH on a seroprevalence survey among our respondents, which will produce a valid estimate of the extent of COVID-19 exposure among people who live with rare diseases. We will conduct linked data analysis once the seroprevalence data become available.
- Finally, we are beginning to design follow-up surveys focusing on vaccine hesitancy and uptake, and to learn about the longer-term impact of the vaccination program on the rare disease community.
About the Survey
In April 2020, a group of RDCRN investigators led by the network’s coordinating center at Cincinnati Children’s Hospital Medical Center undertook a survey of rare disease patients living in the United States. The objectives of the research are:
- To estimate the proportion of rare disease patients who have been diagnosed with COVID‑19 infection
- To describe the characteristics of the COVID-19 presentation and the course of the infection (including treatment) among patients with rare diseases
- To determine whether subgroups of patients defined by sociodemographic variables and geographic location, with particular rare conditions or comorbidities have been affected more frequently or have experienced increased severity of the infection
- To learn about the potential interaction between specific treatment regimens for rare diseases and COVID-19 infection, and specifically whether certain antibiotic, immunosuppressive, or anti‑inflammatory drugs are associated with the frequency of COVID-19 infection and its severity
- To learn about the main concerns that individuals who live with rare diseases and their families have with respect to COVID-19, and determine how the RDCRN can respond by providing information and advice through its network of experts, its consortia, and in collaboration with patient advocacy groups
- To allow follow-up for patients and families who agree to provide contact information, and linkage of information collected in the survey with data maintained by the RDCRN for patients enrolled in RDCRN research studies.
The survey was advertised through the RDCRN with the support of its patient advocacy groups and other partners in the rare disease community. The National Institutes of Health helped disseminate information about the survey through an official press release. The online survey collected variables including patient demographics (race, ethnicity, date of birth, gender), details about COVID-19 infection, and the impact of the pandemic on access to routine care, special food items, and family life, including the impact of stay-at-home orders on mood and behavior, with associated demand for professional support to cope with stress and anxiety.
For more information on the current survey or future plans, please email rd.covid19@cchmc.org.
The Rare Diseases Clinical Research Network (RDCRN) is funded by the National Institutes of Health (NIH) and led by the National Center for Advancing Translational Sciences (NCATS) through its Division of Rare Diseases Research Innovation (DRDRI). Now in its fourth five-year funding cycle, RDCRN is a partnership with funding and programmatic support provided by Institutes, Centers, and Offices across NIH, including the National Institute of Neurological Disorders and Stroke, the National Institute of Allergy and Infectious Diseases, the National Institute of Diabetes and Digestive and Kidney Diseases, the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the National Heart, Lung, and Blood Institute, the National Institute of Dental and Craniofacial Research, the National Institute of Mental Health, and the Office of Dietary Supplements.